Treasure Talk with Bob Evans, News, Rare Coin Articles
Treasure Talk: Episode 5 Part 1
GOLD & RUST
Treasure Talk: Episode 5 Part 1
GOLD & RUST
It is safe to say that we have all experienced rust. Rust is common everywhere. Everyone has grabbed some dirty object only to find their hands soiled with the reddish-brown stains of iron oxide. While it may seem odd to talk about rust in an article about treasure, the reader should realize that the greatest lost treasure in United States history sat for decades in a rusty world.

Hundreds of tons of iron were used in the construction of the S.S. George Law, the steamship that would see its name changed to Central America in June of 1857. At the bottom of the sea, over the course of 13 decades, the massive engines and boilers of the great steamer were transformed into ghostly monuments, reduced to rusty hulks, appearing to melt into the surrounding seafloor. Both chemical and biological agents were responsible for the scene we found in September of 1988.
I don’t want anyone to get the wrong idea. What we saw was the aftermath of a disaster that took hundreds of human lives, an appalling tragedy. My intention has always been to honor the memory of those 425 men who were lost, and the legacy of the 153 survivors, whose lives were changed forever by the Great Storm of 1857. There is a profound spiritual element involved in working on this important site.
As a scientist, there is also the physical reality, and the practical matters that accompany difficult work under challenging conditions, on a deep-sea site that is utterly fascinating and complex, a wonderful manifestation of history as well as natural processes, a shipwreck that shouts obvious facts, but gives up other secrets slowly, if at all.
The S.S. Central America was a wooden ship, constructed of oak and pine.
Marine biologists have a term for what happened when the SSCA sank to the seabed. It was a “woodfall,” usually a word applied to trees that drift out to sea and sink. In the case of the SSCA, it was an enormous woodfall, the equivalent of dozens of trees. From a deep-sea biological resource point of view, in addition to around 600 tons of nutritious wood, it also had lots of small and large nooks and cavities. Even at a depth of 7,200 feet (2200m,) more than a mile below where any sunlight can penetrate, life still proliferates.
Most Americans, for that matter most people, have heard of the Gulf Stream, that great oceanic current that sweeps northeastward up the US Atlantic coast. For centuries it has been an aid (and sometimes a hindrance) to trans-Atlantic travel and commerce. There is another current that flows beneath the Gulf Stream, in the opposite direction, a hidden, undersea river of North Atlantic Deep Water known as the Western Boundary Undercurrent. A thousand miles north of where the SSCA sank, off the coasts of Labrador and Greenland, near to the Arctic Circle, the summer brings extended twilight, nearly 24-hr. sunlight, and so, a huge explosion of photosynthesis. The phytoplankton blooms in the high-latitude summer are important for life on earth. The plankton attracts countless fish and birds, and thousands of whales. The algae in the seasonal soup gush life-sustaining oxygen upward into Earth’s atmosphere, and they saturate the water with dissolved oxygen as well.
This northern water cools, becoming colder and denser than the incoming flow from the Gulf Stream, and so it sinks. It is this cold, nutrient rich, oxygenated mass that constitutes the North Atlantic Deep Water, and that flows a thousand miles southwestward, washing over the S.S. Central America shipwreck site.
Waters of the Gulf Stream and the Western Boundary Undercurrent mix a hundred miles off the southeast US coast, and an enormous dune of drifted sediment has piled up on the seafloor. The Gulf Stream surface teems with billions of microscopic planktonic animals. After they die, their shells slowly rain down. Once these shells reach the bottom of the Gulf Stream they are caught by the cold, south-trending deep water as they descend to the bottom, where they form an expansive triangular mound, 150 miles wide, which has accumulated for eons, becoming the Blake Ridge.
And so, on September 12, 1857, the S.S. Central America sank, fell through the warm waters of the Gulf Stream, passed into the cold depths of the Western Boundary Undercurrent, and impacted the seabed 2200 meters down. It settled onto a flat, slightly sloping field of microscopic shells of plankton, in a region otherwise devoid of any large features.
The sediment is known scientifically as “ooze,” a name which some find amusing. The ooze around the Central America has two main components. Foraminifera, simplified as “forams,” are protozoans, related to the amoeba. But whereas most of us picture an amoeba as an unstructured blob sending out creeping extensions called pseudopods (false feet,) a foram builds a microscopically intricate shell with myriad “foramen” or tiny holes, through which its pseudopods extend to gather food, exchange oxygen, and generally live out its protozoan life. Foram shells are calcium carbonate, the same chemical in limestone, or in certain antacids. It imparts a slightly alkaline character to the near-bottom waters Forams make up most of the ooze, but there is another prominent component, pteropods. Pteropods (pronounced “tare-o-pods”) are snails that swim, as strange as that may seem. In a pteropod, the snail’s foot has evolved into a pair of “wings,” giving the group its name, literally “wing-foot.” In some species the shells have the familiar spirals we associate with snails, but there are also elongate cones, and other elaborate shapes that are perhaps better suited to a hydrodynamic life, swimming around at the surface of the high seas. These snails swim around in the plankton with the forams and all the other near-surface life. Over time, their shells join the mix in the ooze around the SSCA.

So, this was the state of the scene on this particular patch of seabed before the arrival of the SSCA one evening in 1857: a vast expanse of microscopic shells at the upper edge of the Blake Ridge, washed over by the cold and dark, but oxygen and nutrient-rich waters of the Western Boundary Undercurrent. The blanket of shells stretched for miles in every direction. The scene was stable. Unchanging through the ages.
Suddenly, 600 tons of wood and a few hundred tons of iron plopped down in the middle of this setting. For benthic animals, those who live at the bottom of the sea, this was a huge opportunity, the base for a food web to develop, and a hard substrate.
Sessile animals are those who attach to objects and grow on them, like sponges and corals. These animals and many other invertebrates begin life as free-swimming larvae, which drift with the currents until attaching to something hard. It can be anything non-poisonous, a piece of wood, a lump of coal, a gold bar.

The arrival of the wood triggered a feast. Bacteria immediately started to colonize and consume the timbers. Small invertebrates began to graze on the bacteria, and these were in turn eaten by larger species. The nutritious waters provided plenty of food for filter feeders like corals, sponges, and adaptive starfish. The structure of the shipwreck introduced another commodity rare in that vast, flat plain: a third dimension. Animals that crawl could also climb. We found several filter-feeding starfish arrayed high atop the paddle wheel frame and other features that protruded up into the nutritious current.

A complex food web developed within and around this deep-sea oasis, which was unlike any other place for miles around, an opportunity for many species to colonize and thrive.
Along with the wood, the shipwreck introduced another exotic material into the local environment.
Iron.
A brief chemistry lesson:
Iron is the fourth commonest element in the earth’s crust, after oxygen, silicon, and aluminum. So, we know a lot about iron, and with both iron and oxygen so abundant on the planet it is no surprise that rust is as well.
When iron encounters oxygen in air or water it rusts, forming iron oxides or hydroxides. The simplest chemical expression for this is alpha-Fe(OH), the chemical formula for the mineral goethite. The main component of rust is goethite (pronounced roughly by most Americans as “ger-tite,” and named for the great German polymath and poet Johann Wolfgang von Goethe.) It grows into clumpy, earthy deposits on iron surfaces exposed to water or air. Exposure to saltwater adds complexity to the chemical reactions, as the strongly reactive element chlorine becomes involved. Also, rust never passivates the iron, it never forms a protective patina over the underlying metal, allowing mineralization (rusting) to continue. On a large and complex site like the SSCA shipwreck this can result in variations in the local environmental chemistry, from one location on the shipwreck to another, and also vertically, down through the layers of the deposit at the sediment-water interface.

When first recovered, some of the artifacts showed this layering, the changes in chemistry from the thin rust forming in the open water with abundant oxygen, descending into the brownish-red rust at the sediment surface, then deeper into darker and blacker deposits where less and less oxygen is present, and then clear, with no rust forming once deep enough.
It is in this lowest stratum, below the black rust, that we find calcium carbonate adhering to artifact surfaces; “limestone” derived from the microscopic shells that constitute the “soil” where the SSCA settled.
End of chemistry lesson.
What this means for the S.S. Central America shipwreck site is that rust is everywhere. Iron-fixing bacteria proliferate, and they have spread the rust onto almost every exposed surface. Rust bubbles from every iron surface and is redeposited on the ceramics, the coal, and even the gold.
It appears to flow down the sides of machinery and to drip from chains and water tanks like so many icicles or stalactites, like flowstone in cave formations. The team that discovered the shipwreck of R.M.S Titanic dubbed the dangling formations “rusticles.”

Rust was in fact quite an issue for the SSCA treasure.
I have heard it a million times, “Wait a minute! Rust on gold? Gold doesn’t rust, does it?”
No. The rust doesn’t form from the gold. The rust forms on the gold. The rust forms around the gold. It is a surface deposit. In a sense, it is the rock that encases the gold, like the sedimentary rock matrix around fossils.
This is completely unlike tarnish on silver, which involves a chemical reaction between the elements in the air and the silver itself. (Airborne sulfur is a major contributor to tarnish.) So, tarnish is actually a silver mineral, and it represents a change of the original silver surface.
Not so with the rust on the gold of the SSCA. The presence of rust on the gold is due to chemical reactions, aided by biology, but the gold was not involved, except as an inert surface for the deposit.

It is a general tenet of geology that minerals are stable in the environment in which they form. Once removed from that environment they are inherently unstable, the conditions that supported their formation having been disturbed. It is this way with the rust on the SSCA gold.
In an earlier episode of Treasure Talk I wrote about how I merely stabilized the gold when it arrived in my lab aboard ship. It was a fresh catch. Wet and messy. But there was no time for extensive curating; just examine it, catalogue it, make sure it’s OK for now, and pack it up for transport and vault storage.
The gold came up cold, wet, and rusty. Those conditions changed, and the minerals became unstable, as expected. As it sat and dried on the counter next to my shipboard lab sink, the jackets of rust encasing the gold showed that they were changing.
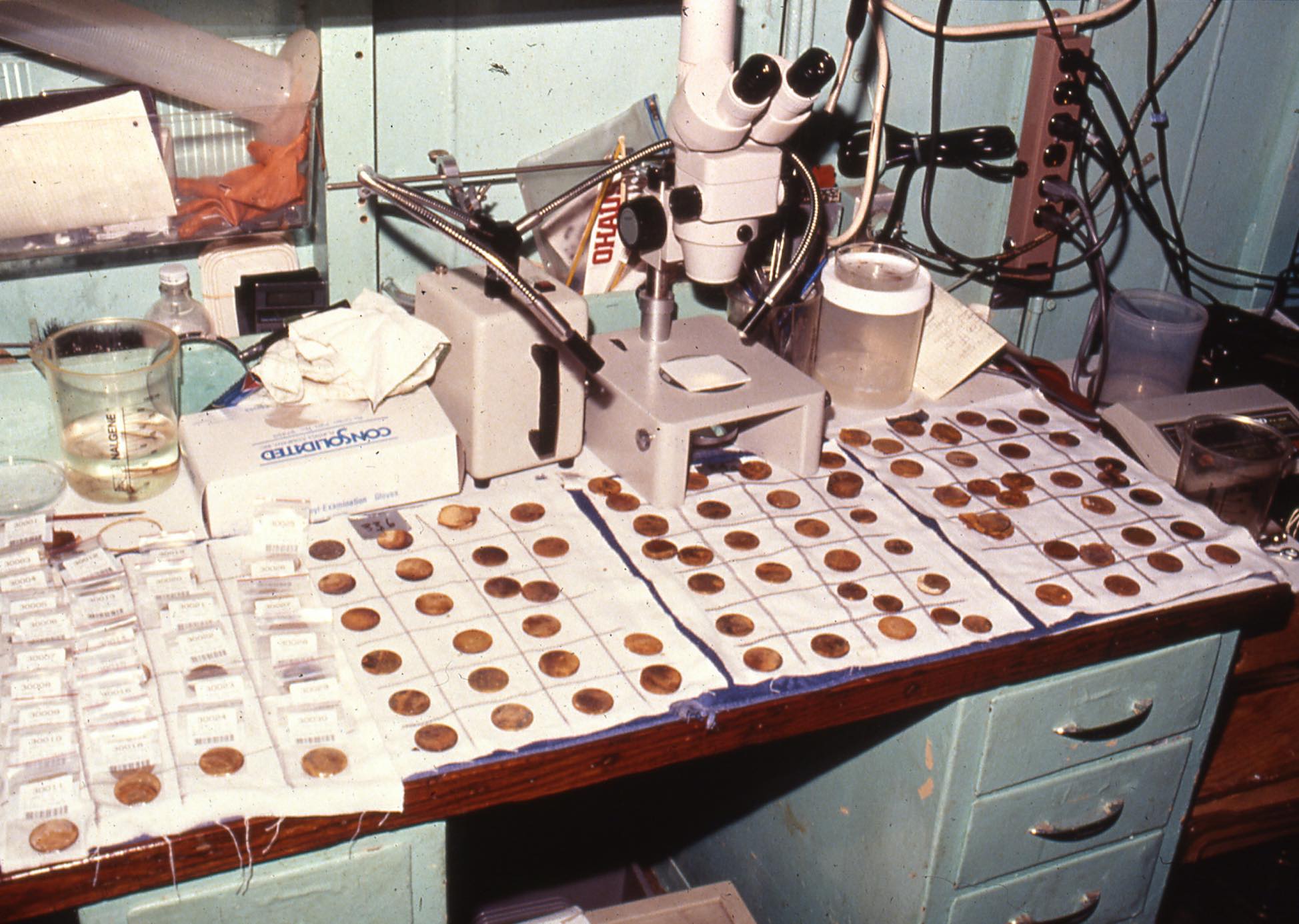
When drying, some of the rust just fell off. Sometimes, I wouldn’t even have to touch it, just having rinsed the piece in the sink and set it out to dry before cataloguing and packing. I would be gazing at a gold bar or coin (because, sometimes you just have to stop and smell the roses,) and a small flake of rust would literally crack and pop off, exposing bright yellow metal underneath.
Maybe the ship’s vibrations contributed, but a couple times I had a strange sense of telekinetic power, like I could remove the rust with my mind, just by staring at it. Cycles of wetting and drying, hydrating and dehydrating the rust, made it expand and shrink, ultimately breaking the physical connection between rust and gold.

Curating over ten thousand pieces of gold, I have now seen every variation of this interface between the rust and the gold.
We found the treasure in 1988, but recovered only a limited amount, preferring to return in 1989 with more sophisticated equipment customized for working on what we had now seen with our own cameras (rather than a hypothetical unknown.) After the 1988 season, I had a few double eagles to work with, a few rusty gold coins. How could I properly remove the rust and reveal what were obviously pretty fabulous coins that were hiding under the dirt? How could I “clean” the gold?
Wait a minute! Numismatists don’t like the term “clean.”
Honestly, coming from the world of science, numismatists don’t like a lot of things. They have high standards, and they are very particular about originality. They are also a very semantic bunch, a fact I find both aggravating and endearing. In a normal world a whole host of synonyms might be useful, in addition to just straight “cleaning.” Washing, rinsing, scrubbing, scouring, polishing, restoring, conserving, preserving, curating.
In the semantic, numismatic world:
“Scrubbing,” “scouring,” and “polishing” are strictly forbidden, because they imply manipulation of the original surface, alteration or movement of metal, so originality is lost.
“Washing” and “preserving” are too general. How did you wash it? What did you do to preserve it?
“Restoring” implies manipulation of surfaces as well, that the restorer has done something to regain the originality, which, let’s face it, cannot be achieved. It also suggests an even worse practice, “doctoring,” in which a coin’s surface or features are treated in a way to either conceal or enhance something about the coin, in other words, to deceive.
This leaves us with “conserving” and “curating.” It could be argued that these are also too ambiguous to be useful. But they sound better than “cleaning,” which is something you do to a dirty bathtub, or a carburetor. They sound more “scientifical.”
I’m fine with being either a conservator or a curator. In my mind, a conservator is a technician, who applies specific treatments or actions to an object to achieve the desired condition. Also, in my mind, a curator is one who cares (from the Latin, curare: to care.) Perhaps it is splitting hairs, but the curator conserves, and catalogues, and studies the objects in question. Curation and conservation are about intent. My intention has always been to do no harm, neither concealing any defect nor enhancing any weakness, and to present a gold coin in as natural and original a condition as possible, to reveal it for what it is.
Somewhere, early in the project, probably after the flood of gold from the 1989 season, I settled on the title Curator to describe my relationship to the treasure. The sudden addition of thousands of pieces to the project’s “collection,” including thousands of the same thing (mint-state but rusty 1857-S double eagles) called for broader curatorial techniques, mostly cataloguing and tracking, rather than the more specific lab work of “conserving” gold.
But as Curator, I was also serving as conservator, and I have ever since. So, call me either.
The public media have repeatedly mangled and conflated all of these “cleaning” terms in describing the role I have played with the SSCA. So please don’t believe it when you read in some newspaper that I restored the coins by polishing them by hand. (!)
Back in the Autumn of 1988 I had no experience with coin conservation. But I had plenty of experience with careful scientific procedures.
In the 70s and early 80s, during my university studies I took a part time job as a technician the Ohio State University geology museum, an institution that boasts a huge, world-class collection of microfossils. Microfossils don’t take up a whole lot of space, so this collection fits adequately within Orton Hall. Several specimens can fit on a microscope slide, unlike a collection of dinosaurs. They are small and unspectacular to most folks, but these tiny remnants of past eons are the key index fossils that allow geologists to figure out the stratigraphy and age relations of the world’s sedimentary rocks. The Orton Geological Museum houses a world-class reference collection. I worked for the museum’s Curator, Professor Stig Bergström, processing limestone specimens collected from all over the world, dissolving crushed samples to isolate and concentrate microscopic, acid-resistant fossils called conodonts, a group of marine organisms that went extinct before the Age of the Dinosaurs.

Microfossils generally represent tiny animals that lived among the plankton, swimming and dispersing over wide areas of the ancient oceans. Fossils of these rapidly evolving animals indicate specific ages for the rocks where they are found. Determining the spatial and vertical distribution of microfossils in the rocks provides data for geologists to figure out the underlying layers and structures within the earth, including the places where we find oil. Most professional paleontologists don’t work on dinosaurs; at least not those with well-funded jobs. They peer through microscopes at tiny fossils to determine where to drill.
So, I had experience with careful scientific procedures, chemicals, and I worked extensively with microscopic detail. Who knew that college work in micropaleontology would be excellent preparation for work as a coin curator?
It was autumn of 1988, and I had a half dozen rusty double eagles we picked up from the edge of the Garden of Gold (our name for the exposed half of the commercial shipment.) My task was to figure out a way to remove the rust from the gold in a manner acceptable to the numismatic community.
In different industrial, business and domestic settings, various cleaning agents are used to attack rust. On ships we use a powerful product called Ospho, for which the main ingredient is phosphoric acid, to prepare metal for painting.
There are various domestic cleaners, including CLR, but that’s acid as well.
Well, those are out. Numismatists don’t like acids, although various acid products have been used judiciously by the scrupulous and less scrupulously by “coin doctors” for years. The decision: avoid acids if at all possible.
Electrolysis:
We consulted with Don Hackman, one of Thompson’s associates from Battelle Memorial Institute, a scientific and engineering thinktank of renown. We tried an experiment with pre-rusted Krugerrands. That was interesting. We duct-taped rusty nails to a couple Krugerrands and dunked them in a beaker of salt water for a month. It worked. Rust on gold achieved. Then we made an electrolytic bath with a solution of water softener (sodium hexametaphosphate) over a waffled aluminum plate in a saucepan over a heat source. This worked, the rust came off the K-rands, but it seemed cumbersome. It would be hard to scale up, and it looked like we were facing thousands of coins.
Meanwhile, (while the K-rands were soaking in saltwater with nails,) I had been doing some parallel research. The Ohio State University has a massive library system. These were the years before the Internet, but within the OSU Libraries, with a little determination, you could find out about almost anything. For those who have no idea, outside of its sports reputation, OSU is huge, with many academic departments of high repute, including the one from which I managed to receive my degree. The Main Library is 13 floors of tightly arranged stacks. OSU also features several departmental libraries with deep holdings.

The Orton Library of Geology is archetypically classic, with large oak tables in the main reading room, and an oak-banistered balcony above with additional stacks. Gazing at the main reading room is a marble bust of Edward Orton Sr., first President of the University, and the Geology Professor (of course), sculpted by Annetta Johnson Saint Gaudens, the sister-in-law of Augustus Saint-Gaudens, the sculptor/designer of the US $20 gold piece, 1907-1933 (as an unexpected numismatic-art connection.)
I did some research at the OSU Libraries, and I couldn’t find anything in the conservation literature, nothing in the world of art or buildings that dealt with the subject of rust on gold. It doesn’t happen in the normal world. I thought maybe on some glorified building somewhere they might have had to remove rust that had bled onto gilt lettering or a golden dome. But no. I guess they probably just use acids and don’t worry about originality, as long as it shines.
So, I came academically home to Geology and Mineralogy, to Orton Hall, where I found there was a computerized search mechanism available behind the Librarian’s desk in a room with some special collections. The Orton Library really is a top geological library, and it didn’t surprise me in 1988, to learn that they were equipped with something called GeoRef, (obviously short for Geology References or similar.) I seem to recall that it was three disks, that could be loaded one by one into the computer and searched separately with key words. I suppose by 2025 standards this was like banging two stones together. But for the time it was a miracle. Titles and abstracts of articles from all the geological and mineralogical journals of every significant language dating back to the 19th century (in many cases) had been loaded into this wonderful device.
I found two articles. One described using a non-acidic solution to remove or alter iron minerals found in soil samples. Interesting. Another described using a similar chemistry to remove iron staining from acid soluble mineral specimens. Beautiful mineral crystal forms, depending on how elaborate, eye-appealing, and rare, can be worth thousands or tens of thousands of dollars. Naturally, since iron is so common in nature, sometimes there are iron stains on these specimens. If the specimen in question is an acid-soluble mineral, the conservator must find a different way to remove such staining without damaging the underlying mineral. Well, that sounded very interesting. Fairly simple, safe, reasonably inexpensive chemistry.
My story, and I have stuck with it for 37 years, is that the solution involves a formulation of three sodium salts in water. One of them is Sodium Bicarbonate, baking soda. And the other two are not. Anyone wanting to know more can figure it out themselves or find out from someone else.

The resultant solution is slightly alkaline, a pH of 7.3. It can be handled with one’s fingers, although I usually use exam gloves. It gives off a slight odor of sulfur, since that is one of the elements involved. When mixed and activated, the solution remains reactive for several hours. I find changing the solution daily is optimal.
After I found the formula in the article at the Orton Library, I needed to test it. I had a few coins from the treasure, but I didn’t want to start with them. There were safer options for initial experiments, no matter how harmless the process looked on paper. I had some lumps of anthracite coal we had recovered from the shipwreck, intended as fuel for the SSCA, now fully coated with rust, just like the treasure. It would not matter much if harm came to a piece of coal. I selected a sort of elongate piece that would fit into a coffee mug. It had rust firmly adhering to the surface, a deposit that would not easily rub off. I filled a 250ml beaker about halfway with the activated solution, put the chunk in the glass, immersing it a little over halfway, so that a part of it sat like an island above the solution, resting against the side. Bubbles began to form on the piece after a minute or two, but no major action immediately. I left it overnight.
When I came back in the morning it was completely successful. The coal was completely shiny black anthracite below the waterline, and unaltered and rusty above.

It worked on coal, now it was time to try it on a coin. I picked out an 1856-S double eagle that I could see was a little dinged up. I could see bag marks through the thin rust over the fields, and part of the coin was more heavily coated, making it a good experimental piece.
I put it in solution in a glass Petri dish overnight. Again, complete success. In the morning, with a little rinsing, I had an 1856-S $20 gold coin in AU condition, looking like it came fresh from the streets of San Francisco in 1857.

This is not that coin, but one just like it: a PCGS AU53 1856-S $20 from the S.S. Central America. Photo courtesy of PCGS TrueView.
In Treasure Talk 5, Part 2 (tomorrow): The Wonders of treasure mineralization.
